The brain is a fascinating organ that connects us to the outside world. It has first to be built, then maintained and eventually shaped by experiences. All these processes rely on the marvellous interactions and coordination between the different cell types that are found within the brain.
What are the cell populations found within the brain?
Neural stem cells are multipotent progenitors that are able to self-renew while producing neuronal and glial progeny. They are in charge of neurogenesis throughout life. They are actively proliferating during embryogenesis to build the nervous system. In addition, during adulthood they are found in a quiescent, mitotically dormant state, from which they can awake to produce new neurons.
Neural stem cell proliferation has to be tightly controlled to produce progeny in adequate number and type, and at the appropriate time. Neurogenesis is ultimately the results of the different inputs, intrinsic, local and environmental, that neural stem cells integrate.
A key player in such regulation is the tailored cellular microenvironment in which neural stem cells reside, or niche. The neural stem cell niche has been mainly described in the mammalian adult brain, and forms a complex architecture made by a diversity of cells: astrocytes, blood vessels forming the blood-brain barrier, microglia which are resident immune cells, and the neurons and neural stem cells themselves.
In particular, the vasculature provides a clear interface between the environment and the brain. Blood-borne, systemic, exogenous factors will first have to cross the physical barrier provided by the tight junctions of the endothelial cells. This blood-brain barrier not only prevents passive diffusion, but can also perform endocrine or paracrine functions targeting neural stem cells in both physiological or pathological conditions.
Glial cells can also modulate neurogenesis. They are closely associated with both the neural stem cells and the vasculature, and can guide newly-born neurons towards their destination. Astrocytes have been shown to locally secrete IGF-I, a metabolic factor neural stem cells respond to to generate new neurons.
It is now clear that each of these different cell types can influence neural stem cell behaviour through a number of signaling pathways, the coordination of which remains to be understood.
In the lab, we are particularly interested in two poorly investigated questions:
Which model?
We are using a simpler yet genetically powerful model, Drosophila melanogaster, to unravel the fundamental cellular and molecular mechanisms behind these processes. The Drosophila post-embryonic brain parallels the organisation of the vertebrate neural stem cell niche. Neural stem cells can be found either in a quiescent or proliferating state depending on the metabolic state of the organism. They are surrounded by various types of glial cells, with a wide range of functions, and by a blood-brain barrier. The Drosophila blood-brain barrier is made of glial cells, and prevents passive entrance of circulating factors by using septate junctions (the equivalent of mammalian tight junctions). It is also able to transmit metabolic signals to the stem cells.
What are the cell populations found within the brain?
Neural stem cells are multipotent progenitors that are able to self-renew while producing neuronal and glial progeny. They are in charge of neurogenesis throughout life. They are actively proliferating during embryogenesis to build the nervous system. In addition, during adulthood they are found in a quiescent, mitotically dormant state, from which they can awake to produce new neurons.
Neural stem cell proliferation has to be tightly controlled to produce progeny in adequate number and type, and at the appropriate time. Neurogenesis is ultimately the results of the different inputs, intrinsic, local and environmental, that neural stem cells integrate.
A key player in such regulation is the tailored cellular microenvironment in which neural stem cells reside, or niche. The neural stem cell niche has been mainly described in the mammalian adult brain, and forms a complex architecture made by a diversity of cells: astrocytes, blood vessels forming the blood-brain barrier, microglia which are resident immune cells, and the neurons and neural stem cells themselves.
In particular, the vasculature provides a clear interface between the environment and the brain. Blood-borne, systemic, exogenous factors will first have to cross the physical barrier provided by the tight junctions of the endothelial cells. This blood-brain barrier not only prevents passive diffusion, but can also perform endocrine or paracrine functions targeting neural stem cells in both physiological or pathological conditions.
Glial cells can also modulate neurogenesis. They are closely associated with both the neural stem cells and the vasculature, and can guide newly-born neurons towards their destination. Astrocytes have been shown to locally secrete IGF-I, a metabolic factor neural stem cells respond to to generate new neurons.
It is now clear that each of these different cell types can influence neural stem cell behaviour through a number of signaling pathways, the coordination of which remains to be understood.
In the lab, we are particularly interested in two poorly investigated questions:
- How does the niche, and its intricate architecture, form?
- To what extent, and for which role, can it remodel under perturbations?
Which model?
We are using a simpler yet genetically powerful model, Drosophila melanogaster, to unravel the fundamental cellular and molecular mechanisms behind these processes. The Drosophila post-embryonic brain parallels the organisation of the vertebrate neural stem cell niche. Neural stem cells can be found either in a quiescent or proliferating state depending on the metabolic state of the organism. They are surrounded by various types of glial cells, with a wide range of functions, and by a blood-brain barrier. The Drosophila blood-brain barrier is made of glial cells, and prevents passive entrance of circulating factors by using septate junctions (the equivalent of mammalian tight junctions). It is also able to transmit metabolic signals to the stem cells.
|
|
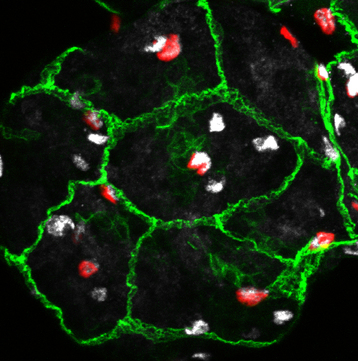
Confocal image of a Drosophila blood-brain barrier in the central brain (projection, early larval stage).
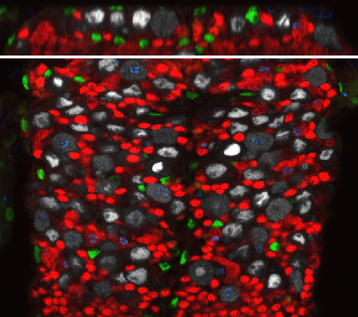
Green, septate junctions. Red, blood-brain barrier glia nuclei. White, glia. Confocal image of a Drosophila ventral nerve chord (one slice, late larval stage).
Top panel, transversal view Bottom panel, ventral view. Green, glia. Grey, neural stem cells. Red, neurons. Blue, phospho-histone 3. |
Which avenues?
We are developing several projects on this cellular model to tackle our favourite questions:
1. Niche morphogenesis (STRUCURES)
A specific subtype of glial cells, the cortex glia, forms an intricate network of membranes spanning the entirety of the nervous system, while encasing each individual neural stem cell and its progeny in a “chamber”. This striking architecture is built progressively, during larval stages, and requires inputs from the neural stem cells (Spéder and Brand, 2018). We are currently using the formation of the chamber network as a paradigm for niche formation (Rujano et al., Nat Commun, 2022).
2. Response of the niche to perturbations (SIGNALS)
We are investigating two avenues. First, we have set up an original model of brain infection by mammalian pathogens in the Drosophila larva (Benmimoun et al., Nat Commun, 2020). It allows us to model both the traversal of the blood-brain barrier, as well as the response of the host cells to infection.
In addition, we are also interested in the adaptation and response of the niche to neural stem cell-derived tumours.
We are developing several projects on this cellular model to tackle our favourite questions:
1. Niche morphogenesis (STRUCURES)
A specific subtype of glial cells, the cortex glia, forms an intricate network of membranes spanning the entirety of the nervous system, while encasing each individual neural stem cell and its progeny in a “chamber”. This striking architecture is built progressively, during larval stages, and requires inputs from the neural stem cells (Spéder and Brand, 2018). We are currently using the formation of the chamber network as a paradigm for niche formation (Rujano et al., Nat Commun, 2022).
2. Response of the niche to perturbations (SIGNALS)
We are investigating two avenues. First, we have set up an original model of brain infection by mammalian pathogens in the Drosophila larva (Benmimoun et al., Nat Commun, 2020). It allows us to model both the traversal of the blood-brain barrier, as well as the response of the host cells to infection.
In addition, we are also interested in the adaptation and response of the niche to neural stem cell-derived tumours.